Clinical Evidence for therapeutic use of Nanobubbles
Applications of Nanobubbles in Neurodegenerative Conditions
In a study performed on cellular models in mice having Amyotrophic Lateral Sclerosis (ALS, also known as Lou Gehrig’s Disease), it was demonstrated that the Nanobubbles of Oxygen have anti-inflammatory and neuroprotective properties.
The authors showed that the injection of Nanobubbled saline solution slowed the disease progression through a significant protection of spinal motor neurons and neuromuscular junctions.
Clinical Studies
Nanobubbles of O2 And CO2 are effective at reducing pain in Peripheral Neuropathy
In a prospective study in 29 patients, Dr. Jeffrey LaMour reports that repeated topical applications of water infused with Nanobubbles of Oxygen and CO2 are effective at reducing pain by at least 50% in 62.1% of patients suffering for Peripheral Neuropathy with no side effects.
Published data show that Lyrica is reducing pain by 50% or more in only 27% and Gabapentin in 40% of the patients treated.
Dr. Jeffrey LaMour presented his report at the TPMA Southwest Foot and Ankle Conference September 12-15, 2019, Frisco, TX.
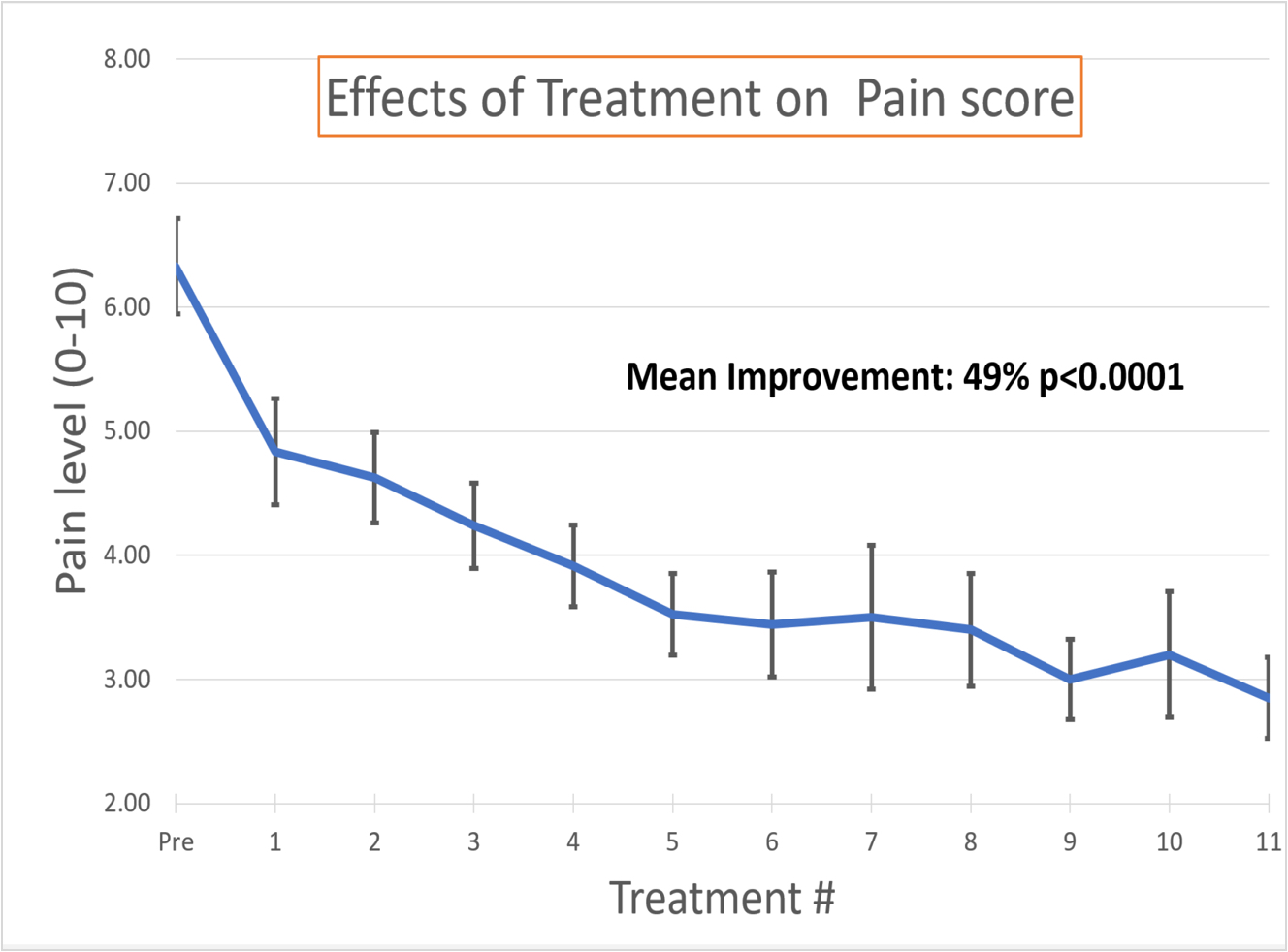
Topical Applications of Nanobubbles infused gel for the treatment of Diabetic Neuropathy
The interim result of a Multicenter prospective IRB-approved (Institutional Review Board) observational study has been presented during the annual meeting of the American College of Foot and Ankle surgery in February 2020. The results show significant improvement of tingling pain, burning pain, numbness, foot sensitivity (measured with Semmes-Weinstein Monofilament Examination) and the global neuropathy disease status evaluated with Toronto Neuropathy Score. Subjects are also reporting a significant improvement of their Quality of Life as measured by the impact of their condition on their general activity, mood, regular work, relationship with other people, sleep and enjoyment of life.
Disclaimer
The statements made on this website have not been evaluated by the Food & Drug Administration. The FDA only evaluates foods and drugs, not General Wellness Products like NoxyPure. NoxyPure is not intended to diagnose, prevent, treat, or cure any disease.